For 20 years, a group of researchers from the Advanced Center for Biotechnology at the University of Yamanashi in Japan has carried out a pioneering and unprecedented experiment that tests the limits. The team, led by Teruhiko Wakayama, a leader in cloning for decades, has managed to create 1,200 mice in series for 58 consecutive generations from a single donor mouse. The results, described by the scientific community as “impressive” and “unrepeatable”, help to understand whether it is possible to maintain a species through cloning and the importance of sexual reproduction in the long-term survival of mammals, including those in danger of extinction.
The research, which was published this Tuesday in the journal , began in 2005 from the study of the genetic material of a female mouse that was cloned using a technique known as somatic cell nuclear transfer, an asexual reproduction process similar to that of Dolly the sheep, in 1997.
The experiment consists of extracting the nucleus of an egg and implanting in it the nucleus of a body cell of the individual to be cloned. In this case, the genetic material was extracted from cells of the cluster that surrounds the oocyte in the ovarian follicle. According to the results obtained, the first generations of clones presented a success rate that gradually increased to the surprise of the researchers until generation 25.
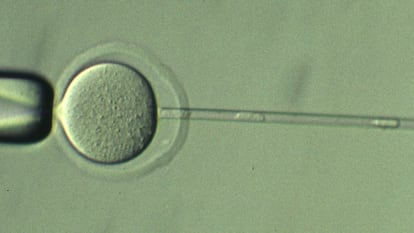
The new mice were born with some minimal genetic alterations that were compensated by their organisms and allowed them to live normally. From there, mutations began to accumulate at an accelerated and harmful rate. Wakayama considers, however, that these limits could still be expanded a little further. “I think we could have pushed the boundaries of recloning even further if we had selected better donors with high cloning success rates,” he responds to this newspaper by email.
The team performed a genome sequencing analysis of the different generations and observed that as mutations accumulated, cloning success rates fell until they were incompatible with life. “The current method of nuclear transfer—which generates three times as many mutations as natural mating—has a fatal flaw that cannot be resolved without developing an entirely new method of nuclear transfer,” he adds.
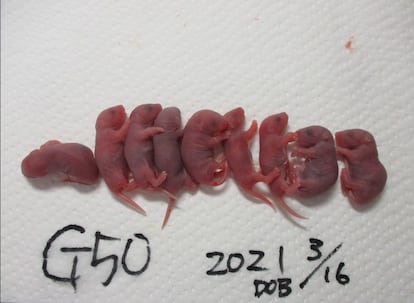
By generation 58, all of the cloned mice died the day after birth. The researchers point out that the mutations accumulated in recent generations exceeded the capacity of their system to compensate for them: in each generation fewer mice were born, the embryos developed worse and the oocytes had more damage, among other findings.
The scientific team’s discovery marks for the first time the limits in cloning in mammals and confirms Muller’s ratchet theory of genetic collapse in asexual reproduction. “These findings highlight the importance of sexual reproduction in mammals. By mixing genetic material from two individuals, sexual reproduction helps repair harmful mutations and ensures the survival of the species, which explains why evolution has favored this mechanism for hundreds of millions of years,” Sagrario Ortega, head of the Mouse Genome Editing Unit at the National Cancer Research Center (CNIO) and not involved in the study, comments to SMC Spain.
The importance of sexual reproduction
Despite the limits of cloning, the mice of the first generations showed normal health and a life expectancy of about two years. Another of the most interesting findings was that although the cloned mice accumulated mutations, they remained fertile and when they reproduced sexually their offspring showed genetic improvements: healthier placentas, larger litters and fewer anomalies, which suggests that sexual reproduction can correct genetic alterations resulting from cloning. “Perhaps sexual reproduction is not so much robust as simply more suitable for the survival of the species compared to clonal reproduction,” explains Wakayama.
“In our own research, even after more than 50 generations of repeated cloning – with mice that had never experienced sexual reproduction – we found no mutations in genes related to pregnancy or childbirth. It is possible that sexual reproduction is indeed more robust than asexual reproduction,” he points out.
Cloning, since Dolly’s birth, has been seen as a promising tool with important applications such as species conservation, biomedical research, and industrial and livestock production. Currently, Wakayama and his team are working on new techniques to clone endangered animals without harming them. “We have managed to create clones from somatic cells present in urine and our current challenge is to create clones from somatic cells present in feces,” comments the Japanese embryologist.
As a researcher specialized in livestock farming, Wakayama explains that his interest in cloning is not theoretical, but deeply practical and seeks to create highly productive animals, such as cows that give more milk or cattle with better meat quality to lower the final cost of food. “Through cloning, I want to allow consumers to buy milk and meat at lower prices,” he says.
However, he acknowledges that the initial idea that a clone is a perfect copy of the original has been left behind. New data shows that cloned animals accumulate mutations over time, which could cause problems in their agricultural use. Therefore, he warns, the next step is to scientifically demonstrate that these mutations do not compromise the health or productivity of livestock before applying these techniques on a large scale. The method he developed in 1998 to clone mice for the first time in the world is still used today.




