In a new study, a team of researchers managed, for the first time, to capture images of an oxygen atom in water, using the fluorescence emitted by excited oxygen atoms before being neutralized by the surrounding water.
Despite being one of the most important elements for life, atomic oxygen continues to contain a surprising number of mysteries, as scientists still do not understand how it reacts in aqueous environments.
This is a crucial point: the oxidizing properties of atomic oxygen are extremely useful in medicine and in industry, so understanding how oxygen reacts in water is essential.
Answering this outstanding question has been practically impossible due to technological limitations.
Previous attempts to measure atomic oxygen in water have failed, as the liquid quickly neutralizes any excited atom before it can be registered.
But now, a team of scientists from North Carolina State University, Princeton and Texas A&M has achieved a significant breakthrough, using a laser technique to femtosecond (10⁻¹⁵ seconds) capable of exciting atomic oxygen and recording the resulting fluorescence before it is extinguished.
The results were presented in an article published in the journal Nature Communications.
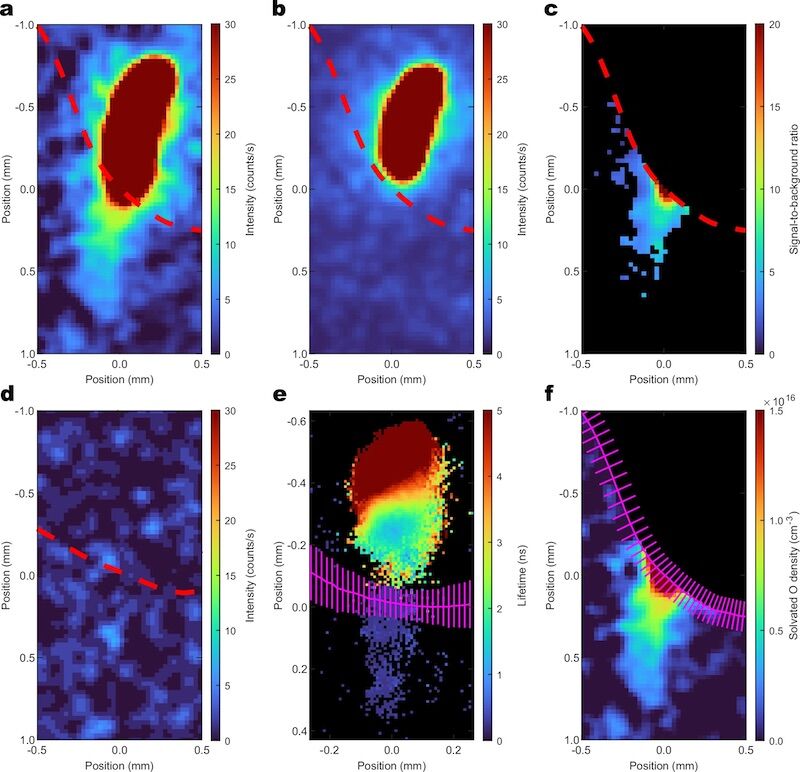
“Measurements show that oxygen atoms persist for tens of microseconds in water, penetrating hundreds of micrometers into the liquid,” the study authors wrote.
“This observed longevity has significant implicationssuggesting the need to reassess existing reactivity models and transport of solvated atomic oxygen”, add the researchers.
To achieve this breakthrough, researchers resorted to a technique known as two-photon absorption-induced fluorescenceor TALIF; and, unlike many scientific processes, this name is actually descriptive.
Scientists measure these individual oxygen atoms by forcing them to absorb two photons simultaneously, which puts them in an excited state. When they return to their ground state, they release excess energy in the form of fluorescence. Measuring this excess energy – or light –it is possible to determine the concentrations of specific forms of an element.
It seems simple, doesn’t it?? The problem is that these excited states don’t last long, as the liquid extinguish them quickly.
This time, however, the use of a femtosecond laser excited the atoms so quickly that the research team had a considerable time window for the atoms. capture before being neutralized.
Myers, B. et al / Nature Communications
Visualizing solvated oxygen atoms with a femtosecond laser
According to , the scientists then compared the fluorescence results with a calibrated xenon density signal, since, as the study authors explain, this presents a practically identical two-photon excitation and fluorescence scheme.
These data were later inserted into simulations to estimate the frequency with which excited atoms collided with water molecules, with the study concluding that there was a estimated total of 10¹⁶ cm⁻³ atoms close to the water surface.
One of the processes that this configuration seeks to understand is the reason why effective branching, i.e. the fraction of excited atoms which each emits a photon.
One of the limitations of the study lies in the implicit assumption that any collision between these atoms excited by the laser and the water always results in de-excitationwhen it is possible for collisions to occur without extinguishing the atom.
Thus, the authors emphasize that the above-mentioned oxygen density values rrepresent, at best, a maximum estimatea, and not an exact number.
As previously mentioned, the study also noted that oxygen can actually persist for tens of microseconds and travel hundreds of micrometers.
Although this distance is unimaginably short for usis significantly higher than scientists predicted, which means that future studies will have to delve deeper into this phenomenon and that existing models on the behavior of oxygen in water will have to be updated.