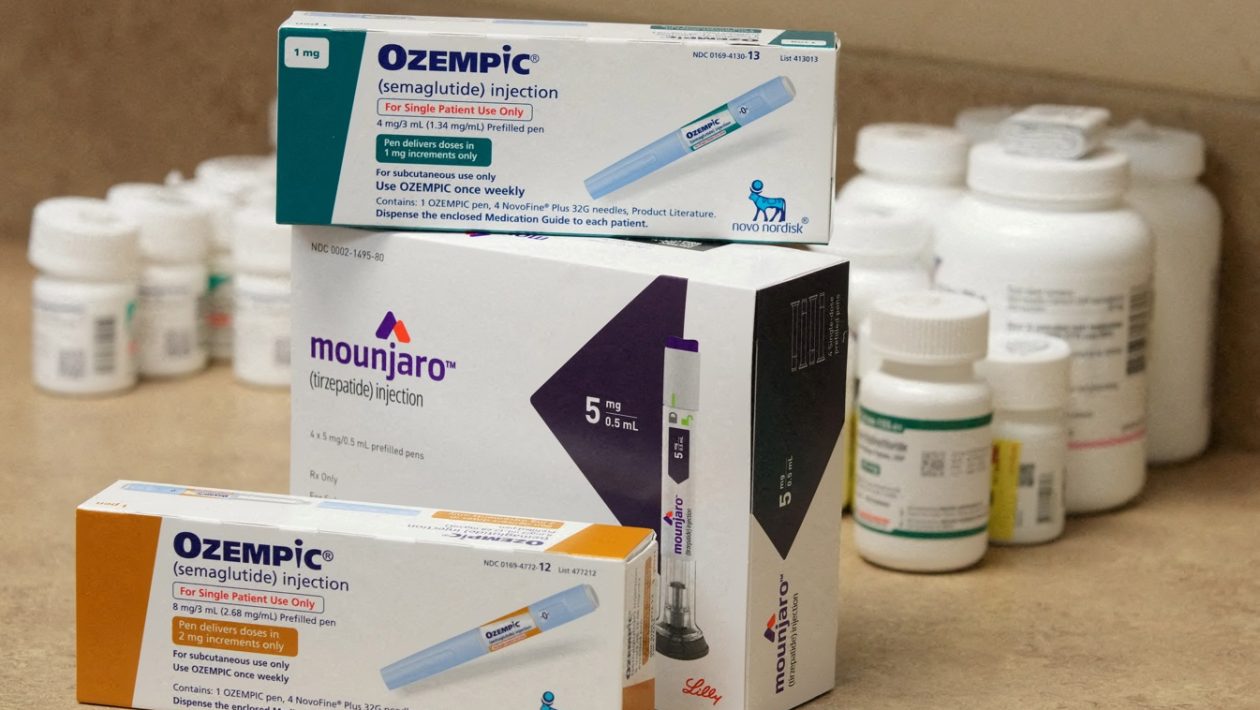
The National Health Surveillance Agency (Anvisa) has 25 requests for analysis for generic semaglutide (Ozempic and Wegovy molecules) and liraglutide in Brazil. Just under half of them, however, have not yet entered the regulatory body’s evaluation stages.
Even before semaglutide’s patent fell, in March 2026, the Agency had already published a notice in the second half of 2025 prioritizing the evaluation of GLP-1 analogue products, a classification given to the drug’s active ingredient. According to the company’s last update, on April 13, .
So far, Anvisa has received 17 requests for analysis of semaglutide. Of them, nine are still awaiting the start of the analysis process, while part of the requests that were already at this stage advanced in the last month, according to information obtained by InfoMoney via the Access to Information Law.
Continues after advertising
An update made by the agency in March reported that eight processes were being analyzed during the period and the deadline for a response to all of them would be the end of April. At the moment, there are: five processes under analysis; one in the requirement stage; one analyzing compliance with requirements; one rejected.
The rejection was made to the Indian pharmaceutical company Dr. Reddy’s, which requested the registration of two injectable solutions of 1.34 milligrams per milliliter. One of the versions had six 1.5 milliliter needles and the other had four 3 milliliter needles. The medicine would be called Embeltah.
Processes in the requirement stage depend on the provision of information to manufacturers so that the analysis can move forward. InfoMoney asked Anvisa whether the deadline for the end of the analysis of the nine processes by the end of April remains, but did not receive a response until the publication of this article.
In the case of liraglutide, only two requests for analysis have not yet been initiated. Two others are in the analysis process and two companies withdrew their requests. Last week, Anvisa announced the rejection of two versions, both from the Indian Cipla, for the Lirahyp medicines.
As InfoMoney published in early April, Novo Nordisk, developer of Ozempic, stopped the patent for semaglutide in Brazil. For now, the Danish company is the only one to produce a medicine of this type in the country.