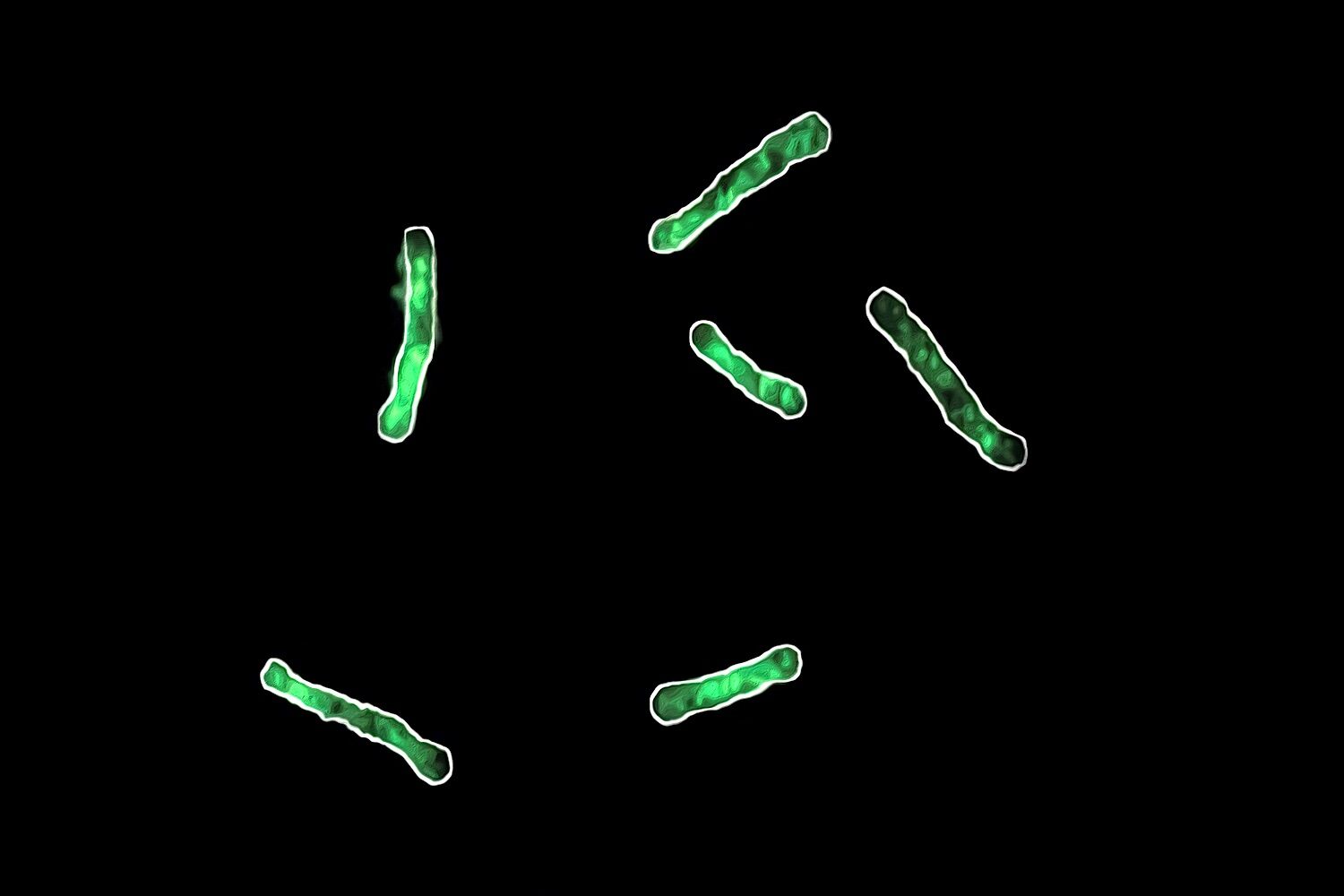
Ashley Moon / Süel Laboratory, UC San Diego
Bacterium Bacillus subtilis with green fluorescence indicating lack of magnesium
A team of North American and Spanish researchers discovered that a strain of bacteria with a mutation that gives it super-resistance to antibiotics loses its ability to survive in situations where magnesium levels are low.
A antimicrobial resistance poses a significant danger to human health. Second, antibiotic-resistant bacteria could cause 2 million deaths per year until 2050.
In a new study, North American and Spanish researchers have now discovered that at least some bacteria pay a high price for its resistance — a cost that we might be able to exploit to combat infections caused by super-resistant bacteria.
“We discovered an Achilles heel of antibiotic-resistant bacteria”, says the molecular biologist Gurol Suelresearcher at the University of California, UC.
“We can take advantage of this cost to suppress the establishment of antibiotic resistance without harmful drugs or chemicals”, adds the researcher.
During the , published in November in Science Advancesresearchers explored why bacteria with resistance factors do not necessarily dominate their relatives not resistant.
The team then discovered a case of protection that comes at a cost for these bacteria, preventing their ability to survive in situations where magnesium levels are low.
“While we often think of antibiotic resistance as an important benefit to bacterial survival, we found that antibiotic resistance ability to deal with magnesium limitation in its environment is more important for bacterial proliferation”, explains Süel.
Magnesium deprivation can neutralize the ability of bacteria to develop. And how Unmutated strains do not share the same flawreducing the key nutrient should not negatively impact the bacteria necessary for a healthy microbiome.
Charged metals, such as magnesium ions stabilize ribosomes — the micro machines in cells that create proteins. Ions also play an important role in using the ATP that powers our cells.
One mutant version of the L22 ribosome in some Bacillus subtilis protects the strain against antibiotics and binds tightly to the charged magnesium atom, leaving less ATP for cellular energy production, explains .
The modeling used by the research team revealed that this physiological cost impedes the ability to grow and spread of the mutated strain, compared to the B. subtilis not mutated.
“Intracellular competition for a finite magnesium reserve you can like this suppress the establishment of a ribosome variant resistant to antibiotics,” the researchers write in their article.
This means that without the pressure of antibiotics, o B. subtilis unmutated is fitter than the B. subtilis resistant to antibiotics.
“We show that, through a better understanding of the molecular and physiological properties of antibiotic-resistant bacteria, we can find new ways to control them without the use of medication“, explains Süel.
“We hope our work can help identify conditions that prevent antibiotic-resistant strains without requiring the development of new antibiotics”, conclude Moon and his team.