Maher El-Kad and /UCLA
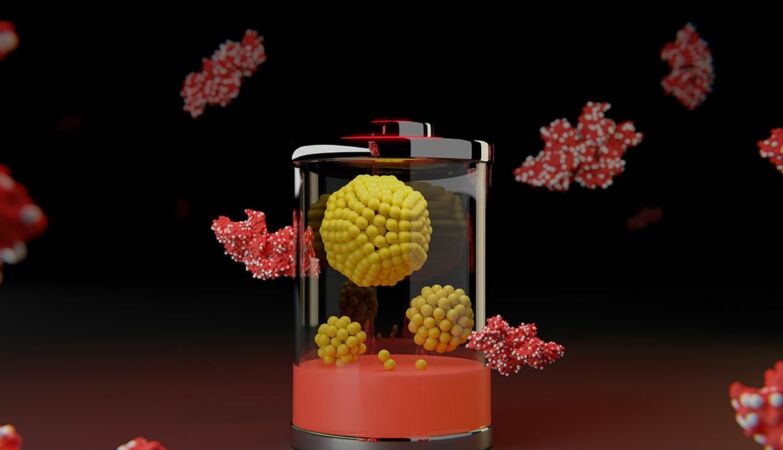
New battery technology: proteins (red) contain tiny clusters of metal (silver). Each yellow sphere in the structures in the center represents a single nickel or iron atom.
Thomas Edison envisioned the potential of electric vehicles long before they became a trend. But during his lifetime, technological advances in gasoline cars pushed his idea, a nickel-iron battery, into the background. Until now.
It’s a little-known fact: in the 1900s, there were more electric cars than gasoline-powered ones on American roads. But the car battery lead acid of the time, courtesy of Thomas Edison, was expensive and had an autonomy of only about 50km.
Edison tried at the time to improve battery technology, and believed that the battery nickel-iron was the future, which promised a range of 160 kma long service life and a recharge time of 7 hoursfast for that era.
Unfortunately, That promise never came true. The first electric car batteries yet suffered from serious limitationsand advances in the internal combustion engine, in addition to discovery of oil wellswhich made gasoline cheaper, tilted the race towards combustion vehicles.
Now an international research collaboration co-led by UCLA was inspired by Edison’s ideasand developed a nickel-iron battery — which may not be able to power automobiles, as the famous inventor intended, but is very suitable for store energy generated in solar parks.
The prototype was able to recharge in just seconds instead of hoursand reached more than 12,000 discharge and recharge cycles — the equivalent of more than 30 years of daily recharges.
The technology was built from tiny metal agglomerates standardized through proteins which were then bonded to a two-dimensional material, made of sheets just one atom thick.
Despite the innovative ingredients, the techniques used in developing the battery, which were presented in a publication in Small magazine, are deceptively simple and economical.
“People often think of modern nanotechnology as complicated and high-tech, but our approach is surprisingly simple and straightforward“, says the biochemist Maher El-Kadyfrom UCLA, and co-author of the study.
“We’re just mixing common ingredientsapplying gentle heating steps and using widely available raw materials”, adds the researcher, from UCLA.
Batteries that receive help from biology
The natural world has provided researchers with some clues. Of particular interest was the process by which animals form bones and molluscs form their hard shells.
Whether the skeletons are internal or external, they are made from proteins which act as scaffolds to collect calcium-based compounds.
The researchers sought imitate this mechanism to generate your tiny nickel or iron agglomeratesexplains Ric Kanerresearcher at UCLA and corresponding author of the article. “We were inspired by the way nature deposits these types of materials.”
“Deposit minerals correctly builds bones that are strongbut flexible enough to not be brittle. The way it is made is almost as important as the material used, and the proteins guide the way they are placed”, explains the researcher.
Isabella Luo / UCLA

Ric Kaner and Maher El-Kady
The team used proteins that are by-products of beef production. The molecules served as templates for the growth of clusters of nickel for positive electrodes and iron for negative electrodes.
Os nooks and crannies in the protein structure folded limited the size of the metal clusters to less than 5 nanometers. This is so small that it would take about 10,000 to 20,000 agglomerates to equal the width of a human hair. The researchers even detected individual atoms of iron and nickel in its electrodes.
The proteins were combined with graphene oxidean ultra-thin 2D material that comes in sheets a single atom thick composed of carbon decorated with oxygen atoms.
Although oxygen can create obstructions that cause the material to act more as an insulator, The process that followed changed everything.
The ingredients were superheated in water and then cooked at a high temperature, causing the proteins carbonize into carbonremoving oxygen from the 2D material and incorporating tiny metallic clusters guided by proteins. The resulting structure was an airgelmade of almost 99% air by volume.
Part of technology’s secret sauce is the surface area — the more exposed, the more space for the reactions behind battery chemistry to occur.
There was a lot of this spaceprovided by the reduced thickness of the graphene airgel and the excess empty space. And the smallness of metallic nano-clusters takes advantage of a fundamental mathematical principle: As objects get smaller, the size of exposed outer surface increases much more than the volume.
“As we go from larger particles to these extremely tiny nanoclusters, the surface area gets dramatically larger,” said El-Kady.
“This is a huge advantage for batteriess. When particles are so small, almost all atoms can participate in the reaction. Therefore, loading and unloading happen much fasterit is possible to store more loadand the entire battery simply works more efficiently”, concludes the researcher.




