according to its announcement, after being informed by the company NUMIL HELLAS SA and taking into account the updated scientific evaluations and recommendations of the EFSA regarding the risk management associated with the cereulide toxin in infants, the DANONE parent company, decided to proceed with a voluntary recall.
What are the lots being recalled?
The recalled batches are as follows, according to a table published by EOF:
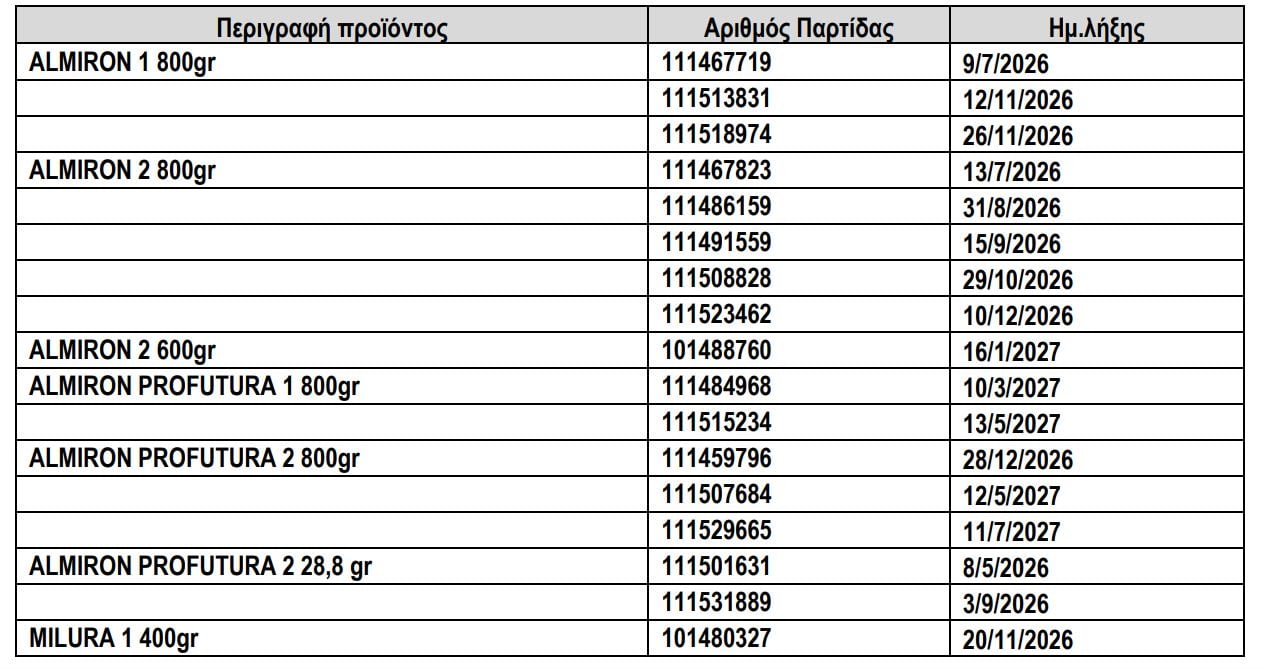
As EOF emphasizes, this decision is issued as a precaution in order to strengthen the voluntary recall of the producer company and extends to the level of the consumer. Consumers who have purchased a product from the mentioned batches are asked not to use it and to return the product to the place of purchase, following the company’s instructions for its replacement.
The company that is responsible for the circulation of the products on the Greek market must, according to the EOF, contact the recipients immediately in order to return the products within a reasonable period of time. The relevant documents are kept for a period of at least five years and are brought to the attention of the EOF, if requested.



