Time was running out for Amanda Sifford—she and her doctors knew it. Amyotrophic Lateral Sclerosis (ALS), the neurological disorder that causes paralysis, was robbing her of her ability to breathe.
In a breathing test, his lung function was at just 48% capacity, a sharp drop from the 86% recorded five months earlier.
“I couldn’t take ten steps and keep breathing,” she said. “I could no longer climb onto a curb.”
Continues after advertising
Sifford, 58, a school psychologist in Cape Coral, Florida, lost 14 family members — including her father and grandfather — to a rare genetic form of ALS, also known as amyotrophic lateral sclerosis or Lou Gehrig’s disease. His symptoms had been developing gradually, but his breathing suddenly dropped.
“It was very scary,” said Nathan Carberry, one of his neurologists at the University of Miami Health System. “I feared we were only talking about a few months to live.”
“I was thinking about death?” Sifford said, pausing to compose himself. “I had already got everything in order.”
It was May 2023, and the Food and Drug Administration (FDA) had just approved the first therapy for a genetic form of ALS, although clinical trial results had not yet proven the drug to be effective.
The drug, tofersen, made by Biogen and marketed as Qalsody, targets the form of ALS that Sifford inherited. So Carberry and Michael Benatar, executive director of the University of Miami ALS Center, rushed to set up a clinic that could administer it.
She began receiving tofersen monthly, through infusions into the spinal canal.
Continues after advertising
It took some time to notice any benefit, and months after starting treatment, she developed spinal inflammation, a serious side effect that required temporary cessation of the medication. But, little by little, his breathing, muscle strength and mobility stopped getting worse — and then started to improve. Results like this are remarkable for a disease that almost always causes continued deterioration and death.

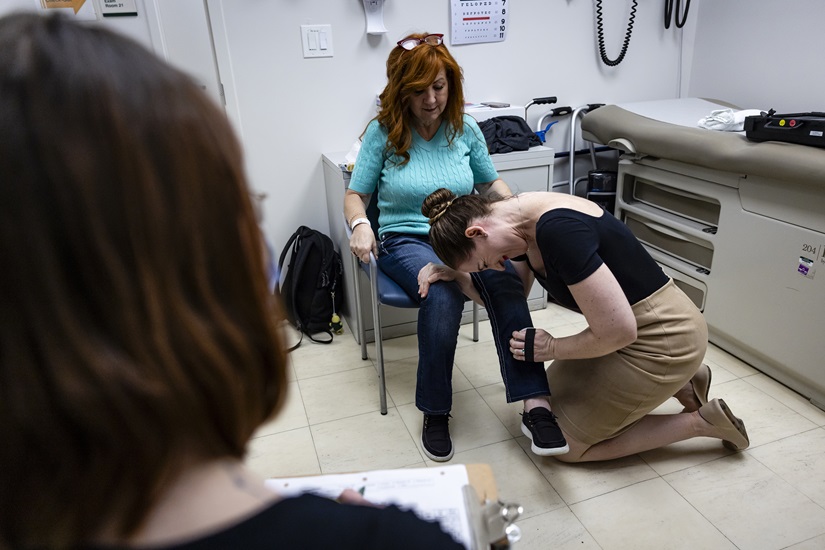
In January, in downtown Miami, she scored 63% on the lung capacity test, the highest score since she started using tofersen in July 2023. “Can you believe this?” exclaimed Anne-Laure Grignon, senior project manager, hugging Sifford in tears.
Tofersen is indicated for only about 2% of ALS patients, those whose disease is caused by a mutation in the SOD1 gene. (About 90% of ALS cases have no known genetic cause; the remainder are due to other mutations.) Tofersen reduces levels of a toxic form of the SOD1 protein.
Continues after advertising
Despite the small number of patients, evidence that some are improving — and that others have stopped getting worse — has generated excitement and optimism among people with a disease that reduces the ability to walk, talk and breathe and usually kills within five years.
Many experts hoped that if tofersen worked, it would only slow the worsening, not stop or reverse it.
“We who care for people with ALS are not seeing improvement,” said Timothy Miller, principal investigator of the tofersen study and a neurologist at WashU Medicine in St. Louis. Patients with the faster-progressing SOD1 form of ALS—the most common variant in North America—often die within a year.
Continues after advertising
When tofersen was approved, the results didn’t show that it worked any better than a placebo. But because it reduced levels of a protein that indicates damage to neurons, the FDA granted conditional approval, requiring Biogen to conduct another study to demonstrate whether the drug could slow ALS.
Now, some evidence has arrived. A study that followed 46 trial participants for about three years found that nearly 20% improved in breathing, strength and functionality.
Although about 75% did not stabilize or improve, their worsening was still slower than expected, said Miller, whose laboratory developed tofersen in partnership with Biogen and Ionis Pharmaceuticals. This was especially true for those who received the drug earlier in the course of the disease, at the beginning of the study, instead of spending six months on a placebo. Among patients with rapidly progressing ALS, those who received tofersen early on lived about three years longer than those who started later.
Continues after advertising
“This shows us that ALS is treatable,” Miller said.
Rickey Malloy, 44, of Hillsboro, Illinois, began taking tofersen weeks after being diagnosed with ALS in July 2023. At the time, Malloy, a longtime plumber, walked with a limp, wore a brace because of foot drop, and needed help climbing stairs and ladders. Today, he says most of these problems have stabilized or improved.
Last year, doctors deemed him healthy enough to undergo knee replacement surgery for an unrelated problem and to participate in a physical therapy clinical trial at WashU Medicine.
“I’m making progress,” he said. “There is no overnight solution and everything is much slower than I would like, but we are making progress.”


For Paula Trefiak of Regina, Saskatchewan, ALS was so common in her family that, as a child, she learned to walk pushing relatives’ wheelchairs and read by holding newspapers to a cousin with the disease. Twenty-six of her relatives died from ALS, including her father, at age 56.
In his early 20s, he began to feel involuntary muscle contractions. An enthusiastic amateur dancer, she had to give up ballet because of balance problems. Later, he had to leave his job as an emergency responder.
She entered the tofersen study shortly after being diagnosed in 2016, but only started receiving the full dose in 2019. Over time, her breathing and strength improved. Today, “I can stand on my toes”, like a ballerina, she said.
She is 44, an age she never expected to reach, as most of her female relatives with ALS died younger.
Recently, her 18-year-old son discovered that he also has the SOD1 mutation, although he still has no symptoms.
“I had that anxiety of thinking: ‘Oh my God, this diagnosis is going to be devastating,’” he said. “But he keeps telling me, ‘Mom, this isn’t the same diagnosis you received. I now have a treatable disease.'”
Despite extensive research, there are only two other treatments approved specifically for ALS: riluzole, which can extend survival by a few months, and edaravone, which can slow progression by about 33%.
Another drug, Relyvrio, was approved in 2022 but withdrawn from the market in 2024 after failing a large study.
Benatar is researching whether tofersen can delay or prevent ALS in carriers of the SOD1 mutation who do not yet show symptoms, but already have high levels of the protein associated with the disease.
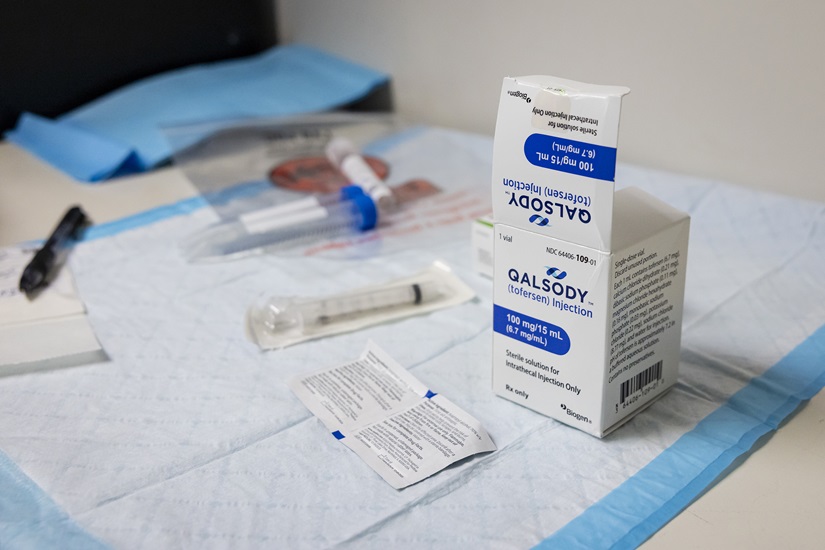

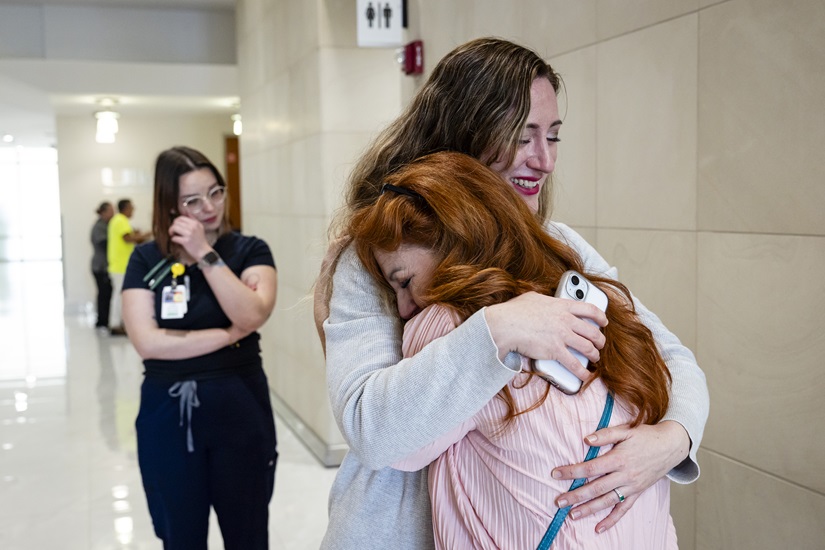
Another big question is whether tofersen could help other ALS patients, those without SOD1 mutations. Miller and Biogen recently began a small clinical study, based on evidence that some patients in the broader ALS set also have misfolded SOD1 proteins, even without the mutation, said Stephanie Fradette, head of neuromuscular development at Biogen. However, according to her, it is still not clear whether these misfolded proteins have any role in causing or worsening the disease.
“It’s far from a won case,” she said. “But it is an important hypothesis to be evaluated.”
The list price for tofersen is $15,500 per monthly dose, making it largely unaffordable without health insurance. Doctors have been fighting battles with insurers who deny coverage to patients. Biogen says it offers copay assistance and a free drug program for eligible patients who can’t afford it.
c.2026 The New York Times Company




