Although Spain is a transplant recipient, every year thousands of people are left without receiving the donated kidney they need. The same happens with other organs. And at a global level the problem is much more serious, with millions of people affected and waiting lists that continue to grow due to the aging of the population. In the United States, seventeen people die every day waiting for a kidney transplant, and every nine minutes one more patient is added to the waiting list. The situation threatens to collapse health systems within a few years. Today, a biomedical advance led by Spanish researchers opens a new way to alleviate transplant waiting lists using human mini-kidneys.
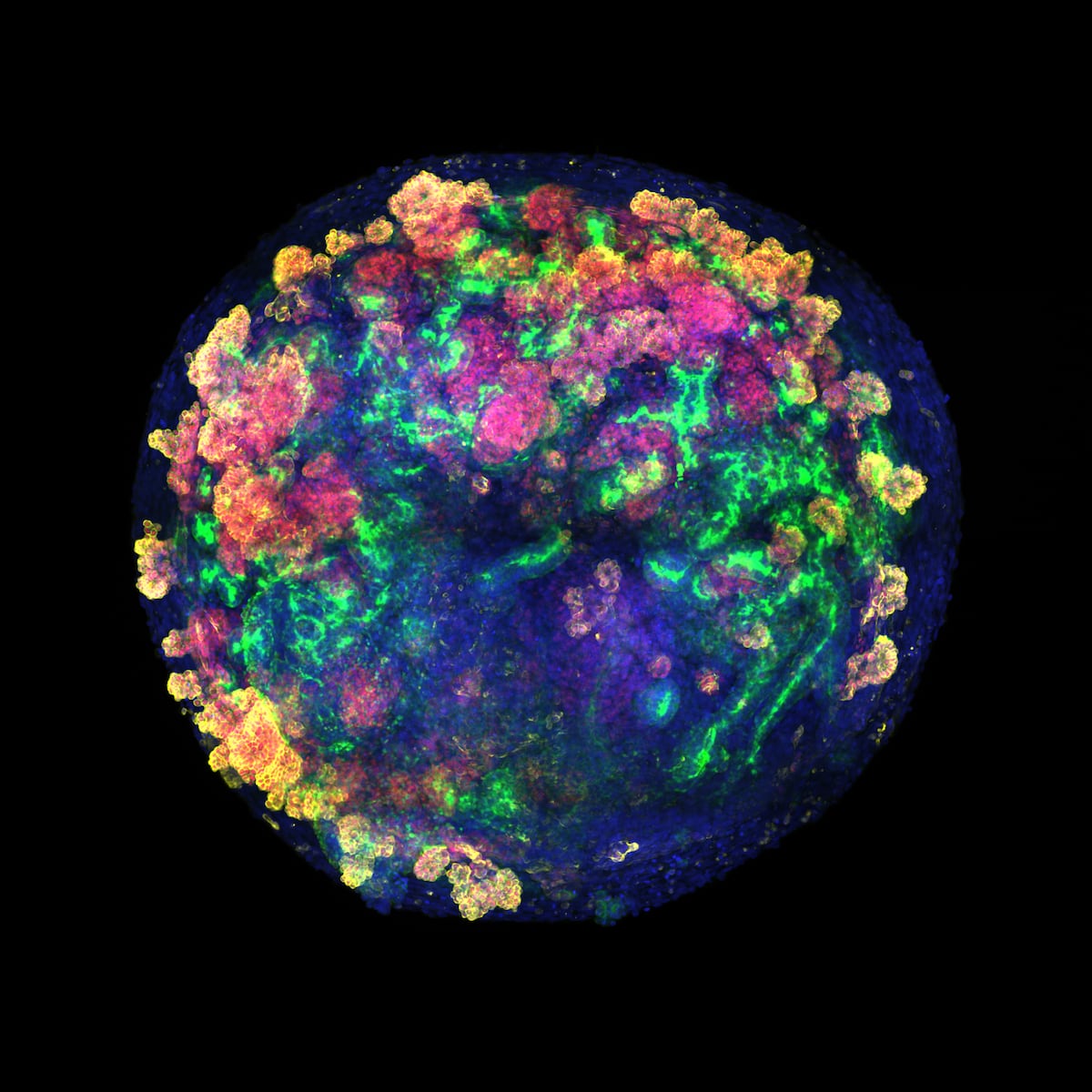
These begin as a mass of a few stem cells, and then grow until they are balls somewhat smaller than the head of a pin. At that time they already have a framework of specialized cells that resembles that of the different organs of the body, a characteristic that makes them ideal for studying diseases or performing tests.
The new work takes a leap to explore whether these structures can also be used for transplants in humans. Researchers have developed kidney organoids from human stem cells, grown them and, for the first time, demonstrated how to produce them in large quantities and cost-effectively, bringing their future application to patients closer.
The work has been directed by the 47-year-old Barcelona biologist Nuria Montserrat, ICREA research professor at the Institute of Bioengineering of Catalonia (IBEC) until just over a year ago, and Research and Universities of the Generalitat of Catalonia.
Before assuming her position in the Government of Salvador Illa, Montserrat had been promoting the use of these cell cultures in regenerative medicine for more than a decade. Now, together with scientists from several Spanish entities, including the National Transplant Organization, and researchers from the University of California, his team has managed to graft human kidney organoids into porcine kidneys for the first time thanks to normothermic perfusion machines, commonly used in the operating room to keep the organ alive and oxygenated outside the body before a transplant. This maneuver allows the integration of human cells into the animal’s organ. For the first time, researchers have re-transplanted these kidneys into live animals from which they had been removed, and have demonstrated their functionality and absence of rejection. The study was published this Friday in the specialized magazine Nature Biomedical Engineering.
The next step, he explains to EL PAÍS, is to test the technique with viscera from human corpses. “Every year more than 1,000 kidneys are discarded that are not suitable to enter the transplant process. Now we have the opportunity to use them for research.” The ultimate goal is to reduce transplant waiting lists and increase the number of kidneys available for graft.
Manufacturing human organoids that are suitable for these uses is extremely complex. “10 years ago,” Montserrat details, “our idea was to make an entire human kidney, from scratch, but it is something very naïve.” “The kidney is one of our most complex organs, with 23 different types of specialized cells,” he adds.
The new kidney organoids are barely visible when they are integrated into the pig organs—they must measure about 200 microns to be able to enter the arteries without clogging them—but they already have 19 different types of specific cells. Thanks to the collaboration with institutions spread across Spain, which are the framework of a future network (development of mini-organs in Barcelona, experiments and transplants to pigs in A Coruña using those manufactured in Zaragoza, and analysis of the immune response at the Carlos III Health Institute in Madrid), this technique has been demonstrated “as a proof of concept.”
The global organ shortage has prompted the use of pig organ meats for human transplants. Until now, one of the most successful cases had been that of Tim Andrews, a 67-year-old American who was grafted with a porcine kidney that had previously been genetically modified to eliminate viruses and make it more compatible with his body. The operation was a success until just a few days ago, when doctors at Massachusetts General Hospital removed it because it was . Andrews lived 271 days with the implant, a world record. The first two patients who received these transplants died shortly after the intervention, and the third was able to carry it for 130 days until his body rejected it. Other living patients have also received experimental heart transplants from these animals, although their effectiveness is still very uncertain. For now, these types of interventions are seen as a bridge with which to gain time until the necessary human organ arrives.
Human organoids are now being added as a new avenue, says , an immunologist at Carlos III and Mount Sinai Hospital in New York, and co-author of the work. “In many cases a patient receives not one, but several kidney transplants due to rejection. These techniques could help make the grafts more compatible with the patient and avoid problems,” he explains. Furthermore, these analogues “can be used in human kidneys that are discarded for transplantation and increase the number of available organs, but they can also be used in kidneys from transgenic pigs to make them more compatible with the recipient,” he details.
It is the first time that quality mini-kidneys have been produced in large quantities and with high reliability, highlights the biochemist, co-author of the study. “Until now, with conventional methods you could create about 40 fabrics of this type. Now, thanks to our system, we can manufacture 30,000 in a single experiment, and we also verify that they are all of high quality,” he points out. There are still years of work ahead, but this advance opens many doors to therapeutic applications, he believes.
The nephrologist, director of the Transplant Organization and co-author of the work, paints the background scenario in which this research is framed. “On any given day, in Europe there are more than 77,000 patients waiting for a kidney for transplant, according to a recent study in 40 countries. They are people doomed to a long treatment with dialysis, with the disruption that this has on their personal and professional development. This is completely transformed by a , which improves life expectancy and is also less expensive than dialysis. This situation is complicated by the progressive aging of donors, who have greater comorbidity. This makes it essential to look for alternative solutions, and Organoids are one of the possibilities for the future,” he explains. The director of the ONT explains that there is already an agreement with hospitals in Barcelona, Madrid, Galicia and Cantabria to begin using human organs discarded in these investigations, expected next year.
Although the application of these human tissues is still a long way off, Domínguez-Gil believes, the doctor predicts two uses of this technology: being able to remove the kidney of a person with kidney disease, treat it with a system similar to that used with pigs and reimplant it; and use this technique to regenerate donated organs that do not meet the requirements for transplant.
The Brazilian doctor was the director of the team that performed the pig kidney transplant on the American patient at Massachusetts General Hospital. The head of kidney transplantation and a professor at Harvard University, who was not involved in this work, praises it as a great first step. “It is an elegant and innovative proof of concept that demonstrates that human kidney organoids can survive and integrate within porcine kidneys for a short period. It is an exciting step towards the fusion of regenerative medicine and xenotransplantation [de animal a humano]. The main challenges in the future will be to achieve long-term viability and demonstrate true kidney function from the transplanted organoids. If the latter is achieved, it could eventually lead to new treatments to restore kidney function in patients with chronic kidney disease,” he highlights.
Marcos López-Hoyos, head of immunology at the Marqués de Valdecilla University Hospital, in Santander, warns that these techniques are still in the research phase in animals, but are still “very interesting.” “Currently, kidneys are already being recovered with ex vivo perfusion methods such as those used in this study. Organoids, although they do not completely reproduce a real kidney, are capable of regenerating it and have enormous possibilities in treatment,” he highlights.